|
Review Article
Comparison of associating liver partition and portal vein ligation for staged hepatectomy (ALPPS) and portal vein embolization (PVE) in the management of advanced liver tumors
1 School of Medicine, University College of Cork, Cork, Ireland
Address correspondence to:
Lim Ai Ying Hannah
Bramley 1, Apartment 10, Orchard Gardens, Dennehys Cross, Cork,
Ireland
Message to Corresponding Author
Article ID: 100006G02JL2021
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Lee JKH, Low KJC, Lim BPY, Hannah LAY. Comparison of associating liver partition and portal vein ligation for staged hepatectomy (ALPPS) and portal vein embolization (PVE) in the management of advanced liver tumors. Edorium J Gastrointest Surg 2021;6:100006G02JL2021.ABSTRACT
Associating liver partition and portal vein ligation for staged hepatectomy (ALPPS) is a relatively new procedure in the management of advanced liver tumors. However, there is limited literature on its benefits over its predecessor—Portal vein embolization (PVE). This review article aims to highlight various aspects of ALPPS and PVE including: (a) patient selection, (b) surgical technique, (c) assessment of volume, growth and function of liver, (d) morbidity and mortality, and (e) oncological outcomes. This could guide surgical clinicians in selecting their approach (ALPPS or PVE) when managing advanced liver tumors.
Keywords: Advanced liver tumors, Associating liver partition and portal vein ligation for staged hepatectomy (ALPPS), Hepatocellular carcinoma (HCC), Portal vein embolization (PVE)
INTRODUCTION
In the past three decades, extensive amounts of research were dedicated toward the field of hepatopancreatic-biliary (HPB) surgery to allow surgeons to securely excise liver tumors [1]. Insufficient volume of the future liver remnant (FLR) was the most common cause of poor postoperative outcomes in patients, leading to post-hepatectomy liver failure (PHLF) [2]. It was in the 1980s when Makucchi et al. [3] first popularized the technique of portal vein embolization (PVE) of a portal venous branch to induce atrophy of the lobe to be resected, and hypertrophy of the contralateral lobe. This enabled the removal of multiple or large tumors located particularly in the right hemiliver and segment IV. This technique was employed by many HPB surgeons and was shown to decrease the incidence of liver failure after right sided hepatectomies [2]. Today, a widely accepted standard care of treatment for patients with an insufficient FLR is an initial preoperative PVE before proceeding on to an extended resection of the liver [4]. However, the inability to reach a desired rate for FLR hypertrophy along with the progression of disease in patients after the initial PVE has led to over 15–20% of patients not being able to have curative liver resection [5],[6].
In 2012, Schnitzbauer et al. [7] first described associating liver partition and portal vein ligation for staged hepatectomy (ALPPS), which was a novel technique developed from traditional two-stage hepatectomies. This procedure of resecting advanced liver tumors occurs in two steps and makes use of the regenerative capacity of the human liver. Unlike PVE, ALPPS is reported to induce greater hypertrophy of the FLR thereby allowing faster progression to the second step. Unfortunately, the procedure has raised major concerns due to numerous reports of its high rates of morbidity and mortality.
This review article aims to discuss the benefits and limitations of each operation (ALPPS or PVE) in terms of: (a) patient selection, (b) surgical technique, (c) assessment of volume, growth and function of liver, (d) morbidity and mortality, and (e) oncological outcomes. This could guide surgical clinicians in selecting their approach (PVE or ALPPS) when managing advanced liver tumors.
PATIENT SELECTION CRITERIA
PVE
Indications
Patients undergoing PVE should not have severe liver dysfunction and should have sufficient predicted FLR. Patients who have underlying steatohepatitis with no metabolic dysfunction would benefit the most from PVE. Patients with systemic diseases like diabetes or who have severe liver dysfunction have limitations on the hypertrophy rate of the liver and subsequent success of the procedure. The probability of hypertrophy of the liver decreases as the volume of the FLR required increases.
There are currently no universal consensus on the ideal minimum FLR, however experts have suggested that in the absence of severe liver dysfunction, PVE may be attempted in patients with a marginal predicted FLR of <20% in patients with normal functioning liver, <30% in patients with non-alcoholic steatohepatitis and <40% in patients with cirrhosis [8]. A wide discrepancy of cut-offs for the FLR had previously existed which was attributed to other factors such as age, systemic or metabolic diseases, background liver disease, and surgeons’ preferences. However, a consensus conference on the resectability of liver metastasis in 2006 has agreed upon a lower cut-off of 20% for minimum FLR in normal functioning livers [9].
Patients with cirrhotic livers are assessed using the Child-Turcotte-Pugh score (A–C). Indocyanine green retention at 15 minutes (ICG-R15) is another factor to be considered in the assessment of patients’ liver function, which determines their suitability for hepatectomy after PVE. In 2015, She and Chok [10] described a protocol from China where patients with Child-Pugh A cirrhosis who had ICG-R15 of <20%; that only a minimum FLR of 30% is needed for right hepatectomy and 35% for extended right hepatectomy. If the patients’ FLRs fall below the stipulated thresholds, then PVE is considered. Shindoh et al. [11] described a major cancer center protocol in the United States for FLR and threshold for hepatic resection in patients with Child-Pugh A cirrhosis. The authors noted that if the patients had an ICG-R15 of <10%, then a minimum FLR of <40% would suffice; and if ICG-R15 is 10–20%, then a minimum of 50% of FLR is required.
Contraindications
Patients who are unfit for major resectional surgery should not be considered for PVE. This includes patients with severe cardiopulmonary comorbidities, portal hypertension and a majority of those who have moderate to severe liver cirrhosis (Child-Pugh B and C) [12]. Additionally, patients who have a generally low performance status should not undergo the procedure. Patients who are unable to tolerate the intervention should not undergo PVE and this includes those with severe sepsis, renal failure requiring dialysis, and patients who have uncorrectable coagulopathy [13]. Patients who have extensive diseases, i.e., extrahepatic disease which cannot be treated curatively or those who are unlikely to achieve margin negative or R0 resection status, are also not recommended for the procedure.
ALPPS
Indications
The use of hepatobiliary scintigraphy (HBS) with single photon emission computed tomography (CT) allows for the measure of regional function of FLR. Patients who undergo ALPPS, regardless of liver quality, should have a cut-off FLR uptake rate of 2.7/min/m2 for safe resection of the liver [14]. Other methods to determine suitability of the liver for ALPPS include measuring volumetric parameters such as total liver volume (TLV), remnant liver volume (RLV), remnant liver volume to total liver volume ratio (RLV/TLV), remnant liver volume to body weight ratio (RLV/BWR) and median volume gain. Associating liver partition and portal vein ligation for staged hepatectomy in patients if RLV/TLV <25% or RLV/BWR <0.5 [15].
In 2014, the first report of the International ALPPS Registry revealed that in 70% of 212 patients who had undergone ALPPS; colorectal liver metastases (CRLM) was an indication for the procedure [16]. The London Health Sciences Centre in 2017 provided several indications for ALPPS in patients who have underlying CRLM [17]. These include: multifocal CRLM requiring extended hepatectomy, predicted FLR <30%, feasible resection (R0 or margin negative), absence of extrahepatic disease (except localized resectable metastasis to lungs), Eastern cognitive oncology group (ECOG) score of 0 or 1, morphology response or absence of progression after systemic chemotherapy, and biological response as seen by reduction of carcinoembryonic antigen (CEA).
Contraindications
The presence of one or more of the following are contraindications for ALPPS: unresectable primary tumors, unresectable tumors in FLR, extrahepatic metastases, portal venous hypertension, or ECOG score ≥ 2 [17]. Other contraindications include: high anesthesiology risk, contraindications to major hepatectomy, unachievable negative margins, or primary unresectable tumors of other locations [18].
Surgical Technique
PVE surgical technique
Percutaneous PVE is performed while the patient is consciously sedated and with local anesthesia. A transjugular or transhepatic approach may be undertaken. Direct transhepatic puncture of portal vein under image guidance by the interventional radiologist is more commonly performed [19]. The transplenic approach is usually performed in patients who have large or multiple tumors where safe trajectory of transhepatic puncture is not feasible [20].
There are two main approaches, either ipsilateral (same side as the intended resection) or the contralateral approach [21]. The contralateral approach was first described by Kinoshita et al. [22] in 1986; and it was later in 1996 when Nagino et al. [23] first described the ipsilateral approach. The current recommendations would be to use the ipsilateral approach in order to avoid access-related complications in the FLR that may compromise resection [24]. The contralateral approach may be a viable alternative if the tumor prevents safe access to the portal branches or if fast-polymerizing ecbolic agents are used.
Numerous embolization agents have been used for PVE including absolute alcohol, cyanoacrylate glue, polyvinyl alcohol (PVA) and coils, and microspheres. Absolute alcohol, although effective, has seen a decrease due to its potentially severe side effects [25]. Cyanoacrylate glue causes a longer lasting occlusion compared to thrombin or gelfoam. PVA with coils are effective occlusives with reduced inflammatory reaction [9]. Madoff et al. [26] have shown that the mean increase in FLR volume using microspheres was much higher compared to large non-spherical particles (69.0% vs. 45.5%). However, the eventual choice of embolic agents will still largely depend on the local expertise, availability, and costs.
The embolization of the ipsilateral portal vein cuts off the blood supply to the area where the largest part of the tumor is being supplied. A portovenogram may be performed to confirm total occlusion of the ipsilateral portal vein, and the patency of the contralateral portal vein and main portal vein. In response to PVE, mitogens such as hepatocyte growth factor (HGF) and epithelial growth factor (EGF) are released which causes regeneration of hepatocytes and hypertrophy of the FLR [9]. Hypertrophy of the FLR peaks at two weeks and the volume of the FLR is reassessed at approximately four weeks after PVE. The surgeon will only proceed to perform resection of the liver when adequate FLR hypertrophy has been achieved.
ALPPS surgical technique
Associating liver partition and portal vein ligation for staged hepatectomy is a 2-step procedure which enables the removal of an extensive part of the liver. The first step involves performing a diagnostic laparotomy to assess for resectability of tumor using intraoperative ultrasound as well as the tumor position with respect to the surrounding vessels. Small tumors in the left lobe may be resected. The ligaments of the liver are dissected, and the right liver lobe is completely mobilized from the caval vein. The right portal vein branch is identified and divided. The portal, arterial, and biliary segment IV branches are also identified and divided. A total or nearly total parenchymal dissection of the falciform ligament may be performed using an ultrasound dissector or Metzenbaum scissors, bipolar coagulation, and hemo-clips [27]. Alternatively, a cavitron ultrasonic surgical aspirator may also be used for the dissection. Parenchymal dissection is performed with division of the middle hepatic vein and preservation of the pedicle to the left lobe. Central venous pressure will be kept at below 5 mm Hg during the dissection. After in situ splitting, the right extended lobe is covered in a plastic bag to prevent formation of adhesions, and the abdomen is drained and closed.
The second step is performed after an interval of 6–12 days. A computed tomography (CT) volumetry is performed, and the procedure may be either completed on the same or following day by relaparotomy. Removal of the plastic covering from the right-extended lobe is performed and the right artery, right bile duct, and right hepatic vein are ligated. Any remaining parenchymal bridges of liver tissue are dissected. Fixation of the left lateral lobe to the anterior abdominal wall via the remnant falciform ligament will be performed to prevent malrotation. A drain is then placed at the resection surface and the abdomen is closed.
Assessment of Volume, Growth, and Function of Liver
Various methods have been used in the clinical assessment of the volume, growth, and function of the FLR. By establishing the differences in volume, growth, and function of the FLR following ALPPS or PVE, it enables surgeons to determine their preference of either procedures in carefully selected patients.
PVE
Estimation of the FLR volume is routinely performed by CT volumetry in patients undergoing PVE [28]. This is essential to determine the suitability of candidates for PVE in order to avoid PHLF. Traditionally, the total non-tumors remnant liver may be estimated using the following formula: (resected volume – tumor volume)/(total liver volume – tumor volume) [29]. However, this method of volumetric assessment often resulted in inaccuracies especially when measuring tumors of larger sizes, and the different liver sizes seen in cirrhotic patients. Furthermore, cumulative mathematical errors often occur when numerous measurements of each tumor is performed in patients with multiple tumors [29]. A separate linear regression total estimated liver volume (TELV) formula was developed based on western adults estimations and is as follows: TELV (mL) = –794.41 + 1267.28 × body surface area (m2) [30]. This has helped to overcome the shortcomings of traditional volumetric assessment of the liver [30],[31]. The standardized future liver remnant (sFLR) is a measurement which allows for a fair comparison of the FLR volume pre- and post-PVE and is derived from the ratio of FLR volume to TELV [32],[33]. FLR growth kinetics of the liver may be expressed in terms of degree of hypertrophy (DH) and kinetic growth rate (KGR) which reflects the regenerative ability of the liver in patients undergoing PVE [34]. Degree of hypertrophy (DH) is defined as the percentage difference between pre- and post-PVE sFLR. Studies have shown that KGR is a better predictor of complications post-hepatectomy compared to sFLR and DH [34]. Kinetic growth rate (KGR) is calculated using the following formula: KGR = DH at first post-PVE volume assessment (%)/time since PVE (days/weeks) at first post-PVE volume assessment. Shindoh et al. [34] also observed a significant increase in FLR and sFLR volume in the first post-PVE volume assessment, where the median DH and KGR were 10.1% and 2.4% per week, respectively. Similarly in another prospective observational study, there was a post-PVE increase in FLR volume from 28.0 ± 6.1% to 38.3 ± 8.5% p <0.001 [35].
In a 2012 systematic review, van Lienden et al. [36] stated that the mean time interval between PVE and follow-up CT volumetric assessment is 25.9 ± 10.1 days and the mean hypertrophy rate post-PVE is 37.9 ± 0.1%. The authors also reported that patients who had undergone PVE had a 96.1% clinical success rate (successful PVE and adequate hypertrophy of FLR for stage two hepatectomy). Of the 70 patients (3.9%) who did not undergo stage two hepatectomy, 51 patients (2.8%) had an insufficient hypertrophy response while 19 (1.1%) patients either had an unsuccessful PVE or a complication preventing resection. The authors concluded that preoperative PVE is an effective method to increase FLR volume with a high clinical and technical success rate.
The functional capacity of the liver is determined by 99mTc-labeled diethylenetriaminepentaacetic acid galactosyl human serum albumin (GSA) or 99mTc-mebrofenin HBS. Total liver function is expressed by the rate of mebrofenin uptake [MUR (%/min/m2)] [32]. The functional share is depicted as a percentage of the ratio between the future remnant volume and the total liver volume. The FLR function is calculated by multiplying MUR with the functional share. Rassam et al. [37] stated that a 69.5 (42.6–108.5)% increase in FLR function of segments 2–3 at three weeks post-PVE was observed [from 1.3 (0.9–1.7) to 2.2 (1.6–2.8)%/min/m2]. Similarly, a 54.7 (30.0–92.6)% increase in FLR function of segments 2–4 post-PVE was also observed [from 2.4 (1.9–3.0) to 3.9 (2.8–4.8)%/min/m2], concluding that there were significant increase in functional share of FLR three weeks post-PVE. This is further evidenced in a different study which showed an increase in function in the non-embolized lobe [from (1.9 ± 0.9) to (3.9 ± 0.8)%/min/m2; p < 0.010] [35].
Interestingly, De Graff et al. [38] stated that the increase in FLR function post-PVE is greater than the increase in FLR volume; concluding that the time to stage two hepatectomy based on FLR function may be shorter than when indicated by volumetric parameters. Similarly, Rassam et al. [37] stated that at three weeks post-PVE, the ratio of increases of functional and volumetric share shifted in favor of function [(51.6% (44.6%–61.1%) vs. 40.8% (34.2%–45.4%)], concluding that time to resection could potentially be shortened.
Many factors impede fair comparison of the outcomes of FLR. Some of these factors include different FLR measurement models, different timing between pre-and post-PVE volumetric and functional evaluation and differences in normal and diseased liver.
ALPPS
Assessing the liver volume in ALPPS is essential to determine if the patient has adequate FLR to progress to stage two [39]. In a 2016 systematic review, Cai et al. [40] stated that ALPPS had a significant increase in FLR volume within 7 to 14 days (range: 47–93%). A more recent systematic review performed by Kang and Schadde [41] in 2017 showed a significant increase in FLR volume in patients who underwent ALPPS within 5 to 14 days (range: 53.7–90.0%). These studies have shown that a rapid hypertrophy of the FLR can be achieved through the ALPPS procedure.
Volumetric assessment including DH and KGR are key measures to assess safety and outcomes of ALPPS [42]. A study has shown that median (IQR) DH was 35 (23–62)% in a cohort of 21 patients with colorectal liver metastases who underwent ALPPS and a median (IQR) time interval between stage one and two was 10 [8],[9],[10],[11],[12],[13],[14] days [43]. In a separate study, DH was also found to be 54 ± 18% when measured one week after stage one of ALPPS [44]. The median KGR in patients experiencing PHLF grade B or C were lower compared to grade A or no PHLF after stage two ALPPS [2.2 (1–7)%/day vs. 6.4 (3–11)%/day; (p = 0.041)] [45]. The study concluded that KGR evaluation pre-stage two of ALPPS is a significant predictor of PHLF.
Testing interstage liver function in ALPPS is key to achieving favorable outcomes [46]. The use of HBS (99mTc-labeled iminodiacetic acid derivatives) together with CT-volumetry performed prior to major liver resection provides a good estimate of FLR function to evaluate the risk of PHLF [14],[47]. The use of CT volumetry alone does not accurately estimate the risk of PHLF in patients undergoing ALPPS. This was shown in a multi-centre study involving 60 patients who underwent ALPPS to compare the differences in functional liver regeneration using HBS and volumetric liver regeneration using CT volumetry. The authors concluded that CT volumetry often overestimated liver function thus resulting in high rates of PHLF [48].
A more recent systematic review in 2017 conducted by Kang and Schadde [41] showed that It is associated with a high incidence of PHLF. The days between stages in ALPPS ranged from 5 to 14 days; with a variable increase in liver function (range: 29.0–52.0%) and increase in liver volume (range: 53.7–90.0%). These findings clearly show that increases in liver volume, which is the principal determinant for progression to stage 2, outweigh increases in liver function. This may explain the high rates of liver failure despite sufficient liver volume post-stage two hepatectomy.
PVE versus ALPPS
Adequate FLR hypertrophy is important to minimize the risk of PHLF. PVE is regarded as the gold standard for the management of advanced HCC, however various studies have reported a 20–30% failure rate which was attributed to postoperative complications such as inadequate hypertrophy or disease progression [49],[50],[51]. There is nearly no failure to proceed with stage two in most, if not all the ALPPS (20% for PVE vs. 1% for ALPPS) [52].
Degree of hypertrophy and KGR are accurate measurements to allow for a good basis of comparison of liver volume and hypertrophy rate for ALPPS and PVE. Croome et al. [50] reported that ALPPS achieved a greater DH compared to PVE [(84.3 ± 7.8%) vs. (36.0 ± 27.2%); p<0.001]. This was supported by a systematic review and meta-analysis conducted in 2019 by Liu et al. [53] where ALPPS had a greater DH compared to PVE (RR: 6.30; 95%CI, 3.97–8.64). This meant that the time to stage two hepatectomy in ALPPS was significantly shorter, thus reducing the incidence of tumor progression, and increasing the probability of achieving tumor-free resection.
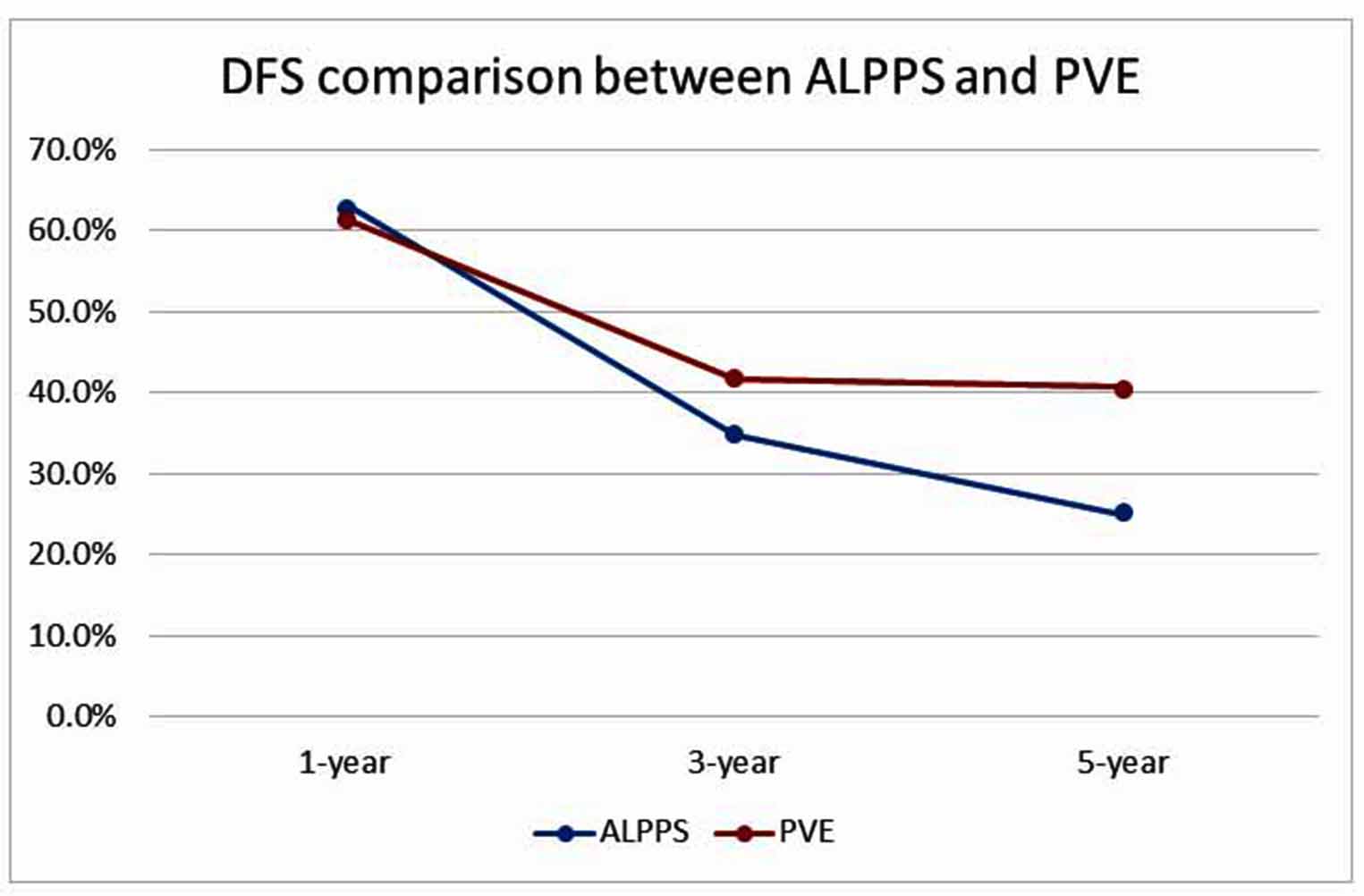
Matsuo et al. [54] stated in their experience that the extrapolated KGR for ALPPS was superior to that of PVE [(14.4 ± 4.8 mL/d) vs. (3.6 ± 2.2 mL/d); p < 0.001]. In 2019, Chan et al. [49] compared the FLR hypertrophy rate between ALPPS and PVE. The authors reported that ALPPS had a greater increment in FLR volume (7.4%/day vs. 1.6%/day), a much faster median time to hepatectomy (7 days vs. 48 days) and a higher resection rate (97.8% vs. 67.7%) compared to PVE. Similarly, another study has shown that KGR was higher in ALPPS compared to PVE [(10.8 ± 4.5%/day) vs. (0.98 ± 0.75%/day) (p<0.001)] [50]. The above evidence clearly shows that DH and KGR is superior in ALPPS compared to PVE.
Liver function is measured pre- and post-operatively in patients undergoing either ALPPS or PVE. Measuring FLR function prior to PVE predicts if patients would have insufficient liver hypertrophy response after PVE; thereby rendering them to be potential candidates for ALPPS [51]. There is currently no literature which compares the postoperative liver function between ALPPS and PVE. Various studies have measured FLR function post-ALPPS using HBS and showed a greater increase of 28–29% over 6–7 days [48],[55], compared to the mean increase in estimated rate of FLR function 4–8 weeks post-PVE in patients with normal liver and chronic liver disease of 16% and 9%, respectively [56]. These evidences favor ALPPS over PVE due to its more efficient increase in liver function over shorter periods of time.
The studies above have shown that ALPPS induces greater increases in growth, volume, and function of the liver compared to PVE. This reduces the chance of tumor progression, and increases the likelihood of achieving complete tumor-free resection in patients undergoing ALPPS. However, there are limited randomized controlled trials comparing liver volumetry and function in PVE and ALPPS. Further higher quality studies should be conducted to better determine the advantages that ALPPS has over PVE in terms of function and growth of the FLR.
Annex 1: Abbreviations and definition table
Morbidity and Mortality
Patients undergoing surgical procedures are at risk of developing postoperative complications that result in morbidity and mortality. Both PVE and ALPPS have differing rates of morbidity and mortality depending on factors such as: initial prognosis of patients undergoing resection, surgeons’ competency on the procedure, and the extent of liver resection. By understanding the incidences and types of morbidity and mortality associated with either procedures, surgeons will be able to make a more informed decision when selecting a surgical approach that suits the needs of their patients. Annexes 2.1–2.3 further summarize the studies which have reported on the morbidity and mortality of PVE and ALPPS.
PVE
Morbidity is defined as postoperative complications occurring within 30 days of surgery, using the Clavien-Dindo classification (Grades I–V). Studies have shown that PVE resulted in a higher postoperative morbidity rate compared to non-PVE [(51–55%) vs. (35–41.1%)] [57],[58]. Huiskens et al. [[57] conducted a retrospective study of all patients undergoing major liver resection in four high volume liver centers in Netherlands from the period of 2000 to 2015 and reported that the total morbidity rate of any complication was higher in the PVE group in contrast to the non-PVE group (51% vs. 35%). Additionally, when comparing major complications (Grades III–V) between the PVE and non-PVE group, the PVE group displayed higher morbidity rates (36% vs. 16%). This was further supported by a systematic review performed by Ironside et al. [59] in 2017 who also reported that the morbidity rate was higher in the PVE group than the non-PVE group (42% vs. 35%).
Mortality rate was also measured in PVE. Huisken et al. [57] reported that PVE had a higher 90-day mortality rate compared to non-PVE (11% vs. 5%). Even when propensity score matching was used, PVE was still proven to exhibit a higher mortality rate (11% vs. 0%). This was also supported by Ironside et al. [59] who reported that PVE induced a higher mortality rate (3% vs. 2%).
The studies above have demonstrated a higher morbidity and mortality rate in the PVE group compared to the non-PVE group. Additionally, Ironside et al. [59] stated that tumor growth was seen in one-third of the patients between the time interval after PVE and before liver resection. This would prevent resection or affect long-term post-resection outcomes. The authors recommend that the use of PVE should only be in patients who are deemed at high risk of PHLF without the operation.
ALPPS
Morbidity in ALPPS is often classified based on the Clavien-Dindo classification of surgical complications (Grades I–V) [60]. In the first landmark study involving 25 patients who had undergone ALPPS for 2-staged extended right hepatic resection, Schnitzbauer et al. [7] reported a total of 50 adverse events in 16 (64%) patients. Of the 50 adverse events reported, 25 were classified as Grade I or II surgical complications that required no intervention, while 22 events were classified as Grade III (requiring intervention or reoperation) or Grade IV (life-threatening including single or multiorgan failure requiring intensive care unit treatment). The remaining three Grade V events (death) occurred in three patients at three different centres. Of note, these three patients experienced a total of 20 adverse events. Perioperative 90-day mortality was also recorded to be 12% (3 of 25 patients).
Several studies later reported variable morbidity rates (range: 36–100%) in patients who had undergone ALPPS[15],[18],[61],[62],[63],[64]. Within these studies, the complications of ALPPS are mainly classified as low morbidity (Clavien-Dindo ≤ Grade III B). The variable morbidity rate was attributed to the low number of participants/patients within each study. Therefore, further research should be conducted with larger samples in order to determine the true morbidity rates of ALPPS.
Scepticism regarding ALPPS was linked to its high mortality rates (12%) in the landmark trial [7]. Further studies have also reported a range of mortality rates (0–29%) [15],[18],[61],[62],[63],[64]. In 2015, Schadde et al. [65] performed an analysis of 320 patients from the International ALPPS Registry and reported that the most common complication is PHLF and the most common direct cause of death is septic shock. Interestingly, other studies conducted on ALPPS have reported a 0% mortality rate [61],[62]. The authors attributed the improvements in mortality to stricter patient selection criteria and clearer refinements in surgical techniques. There is still no firm conclusion that can be derived from the above mentioned articles and that further high quality studies are required to ascertain mortality rates for ALPPS and also its safety and feasibility.
Modifications to surgical techniques have been shown to improve morbidity and mortality rates. Firstly, surgeons should avoid performing routine ligation of bile duct in the diseased liver, as a major complication was bile leaks, leading to increased morbidity and mortality with no significant improvement in FLR hypertrophy rates [66]. Secondly, performing ALPPS using an anterior approach reduces morbidity and mortality associated with the procedure [7]. Lastly, advancements in surgical technology have enabled other minimally invasive approaches to be incorporated into ALPPS. This includes the invention of tourniquet-ALPPS [67], laparoscopic mini-ALPPS [68], laparoscopic ALPPS[69] and partial ALPPS [70], that also improve morbidity and mortality rates. These modifications however are still relatively new and there is still no convincing evidence to back them.
PVE versus ALPPS
Morbidity rates of PVE and ALPPS may be classified using the Clavien-Dindo classification (Grades I–V); where overall morbidity rates are Grades I–V and major morbidity rates are Grades III–V. In the 2019 systematic review and meta-analysis, Liu et al. [53] compared the safety of ALPPS versus PVE and reported that the overall morbidity rates were greater in ALPPS compared to PVE [(25% vs. 21%); RR: 1.37, 95% CI, 0.84–2.21]. Similarly, Shindoh et al. [71] reported that the overall morbidity rate and major morbidity rates were higher in the ALPPS group compared to the PVE group [(64% and 40%, respectively) vs. (57.7% and 32.7%, respectively)]. The authors also noted that ALPPS had greater incidences of bile leak [International Study Group of Liver Surgery (ISGLS) grade B or C], sepsis and relaparotomy for postoperative complications compared to the PVE group [(24.0%, 20.0%, and 28.0%, respectively) vs. (5.8%, 0.0% and 2.9%, respectively)].
Conversely, in a single centre study in Hong Kong, Chan et al. [49] reported that there were no differences in overall morbidity rates between ALPPS and PVE (20.7% vs. 30.4%; p = 0.159). Bednarsch et al. [43] classified major morbidity as Grades II–V. The authors reported that although ALPPS had a higher major morbidity rate compared to PVE (64% vs. 38%), the results were not statistically significant. The morbidity rates associated with ALPPS compared to PVE are controversial and further studies with greater sample sizes still have to be conducted in order to make a fair comparison between either procedures.
While ALPPS has a clear superior completion rate compared to PVE, there is still controversy with regard to the mortality rates. Chan et al. [49] stated that there was no difference in mortality rates between ALPPS and PVE (6.5% vs. 5.8%; p = 1.000). The authors stated that there were no clear risk factors for mortality identified on multivariable analysis for ALPPS; however the presence of refractory ascites post-stage I was regarded as a contraindication for stage II operation. Conversely, Liu et al. [53] reported that the adjusted mortality rates were higher in ALPPS compared to PVE in three studies [10% vs. 5%; (RR: 2.26, 95% CI, 0.88–5.80)]. The authors also stated that patients with signs of liver failure [e.g., Model of End stage Liver Disease (MELD) score >10] post-step one of ALPPS should not undergo step two; and that older patients (>60 years) have worse prognosis as well.
Based on the studies above, the results of mortality and morbidity rates of ALPPS still remain controversial. Among the studies, Liu et al.’s systematic review provided a greater level of evidence which supports ALPPS having superiority over PVE in terms of DH of FLR but at a cost of higher rates of mortality and morbidity. A limitation of the systematic review, however, is the lack of randomized controlled trials comparing ALPPS and PVE which makes it difficult for us to make a fair comparison between either approaches. Therefore, we recommend further higher quality studies to be conducted in this area.
Oncological Outcomes
PVE and ALPPS are routinely used to induce liver hypertrophy prior to major liver resection in patients with hepatic malignancy. This allows for a larger future liver remnant, and hence an increased number of patients suitable for resection. However, there are concerns that preoperative induction of liver hypertrophy may promote tumor growth and increase recurrence rate. Annexes 3.1–3.3 further summarize the studies which have reported on the oncological outcomes of PVE and ALPPS.
Annex 2: Summary of studies reporting on the mortality and morbidity
Annex 2.1: Mortality and morbidity (PVE)
Annex 2.2: Mortality and morbidity (ALPPS)
Annex 2.3: Mortality and morbidity (PVE vs. ALPPS)
Annex 3: Summary of studies reporting on the oncological outcomes
Annex 3.1: Oncological outcomes (PVE)
Annex 3.2: Oncological outcomes (ALPPS)
Annex 3.3: Oncological outcomes (PVE vs. ALPPS)
PVE
Disease progression after PVE is defined as the percentage change in total tumor volume. Although PVE induces hypertrophy of the FLR, it has also been shown to promote a compensatory increase in portal flow to the tumors, increasing tumor volume through the upregulation of growth factors and worsening disease progression [72]. Tumor unresectability often results in patients being unsuitable candidates for stage two hepatectomy [73].
Several studies reported that 11–18.5% of patients who underwent preoperative PVE could not undergo resection due to disease progression [71],[74]. Other studies done defined disease progression as tumor growth rate (TGR) measured by CT volumetry. In a study reported by Hoekstra et al. [74], the median TGR for patients who underwent PVE was higher compared to non-PVE patients (0.53 mL/d vs. 0.09 mL/d). Disease progression may also be measured in terms of the development of new lesions in the FLR or extrahepatic disease. Simoneau et al. [73] reported in 2015, where 78.1% of patients who had undergone PVE had developed disease progression (66% tumor progression and 12.1% new lesions in the FLR).
The oncological outcomes of patients who have undergone PVE may also be measured in terms of disease-free survival (DFS). Disease-free survival is the percentage of individuals in the treatment group who are free of signs and symptoms of a disease, and is measured from the date of liver resection until the date of diagnosis of recurrence or last follow-up visit. A systematic review conducted by Ironside et al. [59] in 2017 reported that the DFS was lower in the PVE group compared to the non-PVE group (median: 15.2 months vs. 21.7 months). A multi-center analysis performed by Huiskens et al. [57] in 2018 showed that there was no significant difference when comparing 1 and 3-year DFS in a matched cohort of PVE and non-PVE groups [(44% vs. 44%) and (9% vs. 16%) respectively; p = 0.776].
Overall survival (OS) rate may be used to measure oncological outcomes in patients who had undergone PVE. Overall survival is the percentage of people in a study or treatment group who are still alive for a given period of time after diagnosis and may be measured from the date of PVE until the date of death or date of last clinical follow-up or from the date of diagnosis of liver metastasis until the date of death. Whicherts et al. [58] reported that PVE was associated with a lower 3-year OS as compared to non-PVE groups (44% vs. 61%; p = 0.001). Interestingly, Huiskens et al. [57] reported no significant difference between 5-year OS when comparing PVE and non-PVE groups (14% vs. 14%; p = 0.866). Similarly, a systematic review and meta-analysis performed by Giglio et al. [75] reported that PVE has no significant effect on 3- and 5-year OS between PVE and non-PVE groups.
We observed that the PVE group had a worse oncological outcome and prognosis compared to the non-PVE group in terms of OS and disease progression [58],[71],[73],[76]. However, limitations of the study include the lack of propensity score matching which reduces selection bias between the PVE and non-PVE group. Patients who underwent PVE had a greater tumor load or more extensive metastasis, which explained the significantly lower DFS and OS, and higher disease progression. In a study conducted by Huiskens et al. [57] in 2018, propensity score matching based on oncological parameters was performed to reduce selection bias, resulting in similar DFS and OS when comparing PVE and non-PVE groups. Although there is greater disease progression post-PVE, it does not seem to have a significant impact on oncological outcomes in the long term. Instead, it proves to have short-term benefit by rendering patients with CRLM to be able to qualify for resection.
ALPPS
Associating liver partition and portal vein ligation for staged hepatectomy is associated with a greater increase in kinetic growth rate (KGR) enabling adequate hypertrophy of the FLR compared to PVE. Disease progression is not relevant in patients undergoing ALPPS due to the shorter time interval between stages 1 and 2, thus rendering them suitable candidates for stage two hepatectomy. In a systematic review performed by Dimitrios et al. [77] in 2017, numerous studies had reported that most of the patients who had undergone ALPPS were able to proceed to the stage two hepatectomy (range 83.3–100%). In contrast, the PVE procedure had shown a smaller proportion of patients proceeding to stage two hepatectomy (range 63.3–100%), owing to disease progression and inadequate hypertrophy of the FLR. This portrays a higher dropout rate in the PVE cohort.
Oncological outcomes of ALPPS may be measured in terms of DFS. Disease-free survival is the percentage of individuals in the treatment group who are free of signs and symptoms of a disease, and can be measured from the date of last hepatectomy until the date of first recurrence or death. Ratti et al. [78] reported in 2015 that the 1-year DFS in the ALPPS group was 67%. Interestingly, Adam et al. [79] reported in their experiences that the 1-year DFS in the ALPPS group n = 17 was 0%. The authors postulated that the low DFS could have been attributed to the more aggressive and accelerated recurrence of tumors following ALPPS, in which the increased KGR might have triggered residual tumor progression. The greater variability in the DFS among these studies could be due to the small patient cohort, which reduces the confidence interval of these studies.
Another measure of oncological outcomes in ALPPS is OS. Overall survival is measured from the date of liver metastasis until the date of death. Björnsson et al. [72] reported that the 1-year, 2-year, and 3-year OS of patients who had undergone ALPPS was 96%, 73%, and 60%, respectively. Ratti et al. [78] had also reported that the 1-year OS of the ALPPS group was 92%. Adam et al. [79] in 2016 also reported that the 2-year OS of the ALPPS group was 72%.
We observed that the dropout rate and progression to stage two hepatectomy is higher in PVE as compared to ALPPS, owing to disease progression. As ALPPS induces a greater KGR and shorter time to achieve sufficient hypertrophy of the FLR, a greater proportion of patients are selected to proceed to stage two hepatectomy. We also observed that the studies for ALPPS contained small patient cohorts, which might have resulted in the greater variability in DFS rates in these groups.
PVE versus ALPPS
Disease-free survival and recurrence free survival (RFS) are important components when comparing the oncological outcomes of PVE and ALPPS. Figure 1 compares the DFS between ALPPS and PVE. Chan et al. [49] reported in 2019 that the 1-, 3-, and 5-year DFS in the ALPPS and PVE group were not significantly different [(63.2%, 34.9%, and 25.0%) vs. (61.4%, 41.8%, and 40.7%), respectively; p = 0.267]. Interestingly, Bednarsch et al. [43] reported in 2020 that the 3-year RFS in the ALPPS group was not significant compared to the PVE group (43% vs. 8%; p = 0.05). Furthermore, the median RFS for ALPPS was 19 months, compared to the PVE group of 10 months. Although the ALPPS group appeared to have a better RFS, the authors have stated that there was no significant difference in oncological outcomes between ALPPS and PVE. The studies listed above have shown that the DFS of ALPPS and PVE are comparable.
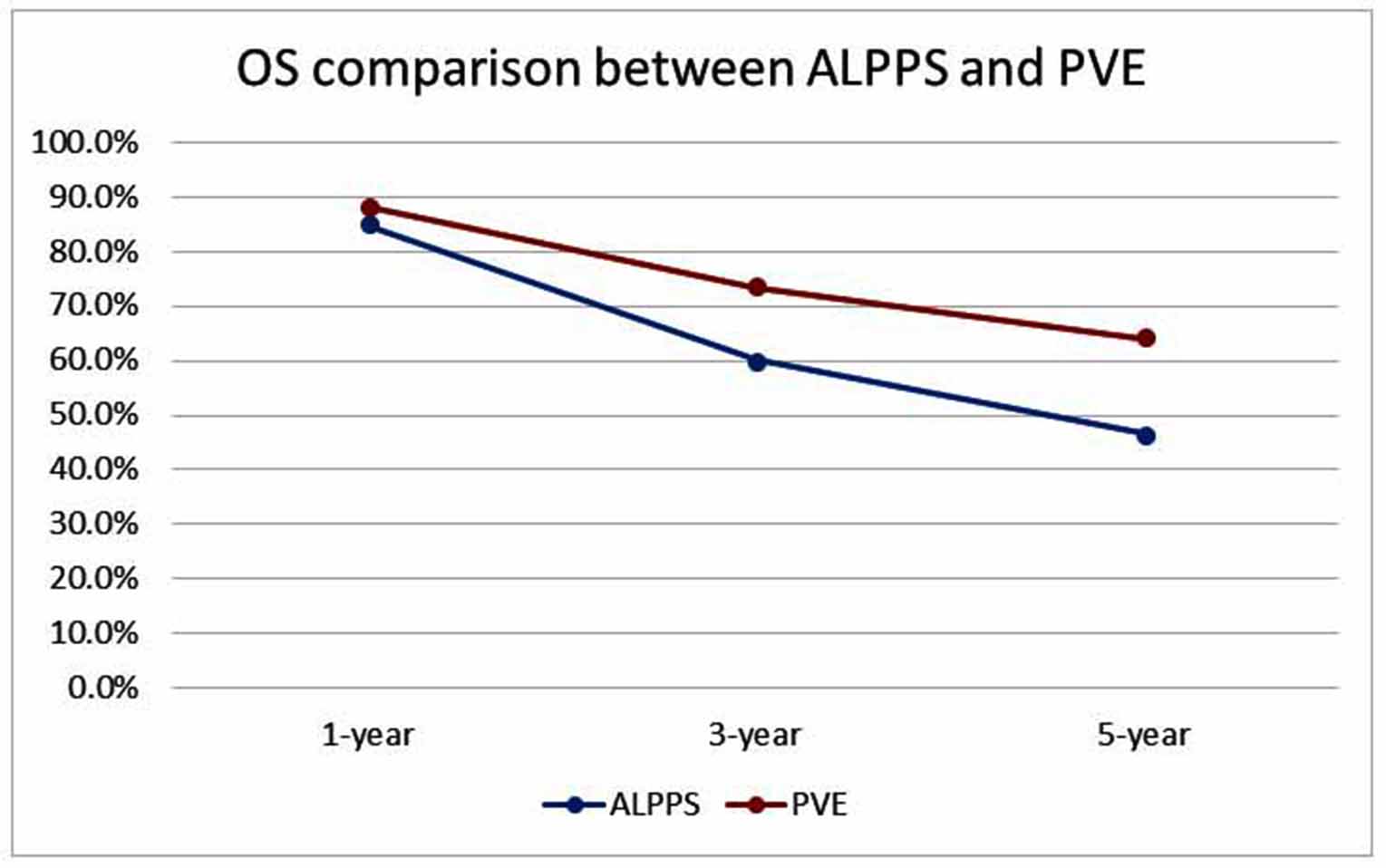
The OS was also comparable between ALPPS and PVE. Figure 2 compares OS between ALPPS and PVE. Chan et al. [49] reported that there was no significant difference in the 1-, 3-, and 5-year OS of the ALPPS and PVE group [(84.7%, 60.2%, and 46.8%) vs. (88.2%, 73.5%, and 64.1%); p = 0.234]. The authors had also stratified both groups based on tumor staging, and concluded that the OS was comparable between both groups. This was supported by Bednarsch et al. [43] who reported that the median overall survival duration between ALPPS and PVE groups was 28 months versus 34 months. Additionally, the 3- and 5-year OS was not significantly different between the ALPPS and PVE group [(37% and 37%) vs. (44% and 33%); p = 0.297].
In a systematic review performed by Moris et al. [77] in 2017, a smaller proportion of patients who had undergone PVE were able to proceed to stage two hepatectomy compared to ALPPS [(range 63.3–100.0%) vs. (range 83.3–100.0%)], due to progression of disease and inadequate hypertrophy of the FLR. This portrays a higher dropout rate in the PVE cohort. This could be explained by the tumor progression as a result of a longer time interval post-PVE, the presence of new tumor lesions in the FLR, as well as manifestations of extrahepatic disease. This also suggests that ALPPS offers a higher chance of a complete resection in the management of advanced liver tumors.
These results have shown that neither ALPPS nor PVE have superior oncological outcomes. Various studies reported that the DFS and OS of both procedures were comparable [43],[49]. Patients undergoing ALPPS would achieve a higher resection rate compared to PVE mainly due to disease progression. Further studies are required to determine the short and long-term oncological outcomes in a larger patient cohort.
CONCLUSION
ALPPS may be considered as a viable alternative procedure to PVE in the management of advanced liver tumors. ALPPS is associated with greater growth and hypertrophy of the FLR, higher rate of completion of stage two resection, and comparable short and long-term oncological outcomes compared to PVE, albeit at the cost of higher rates of morbidity and mortality. As there is still limited evidence to support the superiority of ALPPS over PVE, we recommend that further higher quality studies should be conducted for a better assessment of the benefits of ALPPS over PVE.
REFERENCE
1.
de Santibañes E, Clavien PA. Playing Play-Doh to prevent postoperative liver failure: The “ALPPS” approach. Ann Surg 2012;255(3):415–7. [CrossRef]
[Pubmed]

2.
Clavien PA, Petrowsky H, De Oliveira ML, Graf R. Strategies for safer liver surgery and partial liver transplantation. N Engl J Med 2007;356(15):1545–59. [CrossRef]
[Pubmed]

3.
Makuuchi M, Thai BL, Takayasu K, et al. Preoperative portal embolization to increase safety of major hepatectomy for hilar bile duct carcinoma: A preliminary report. Surgery 1990;107(5):521–7.
[Pubmed]

4.
Kim D, Cornman-Homonoff J, Madoff DC. Preparing for liver surgery with “Alphabet Soup”: PVE, ALPPS, TAE-PVE, LVD and RL. Hepatobiliary Surg Nutr 2020;9(2):136–51. [CrossRef]
[Pubmed]

5.
Nagino M, Kamiya J, Nishio H, Ebata T, Arai T, Nimura Y. Two hundred forty consecutive portal vein embolizations before extended hepatectomy for biliary cancer: Surgical outcome and long-term follow-up. Ann Surg 2006;243(3):364–72. [CrossRef]
[Pubmed]

6.
Abulkhir A, Limongelli P, Healey AJ, et al. Preoperative portal vein embolization for major liver resection: A meta-analysis. Ann Surg 2008;247(1):49–57. [CrossRef]
[Pubmed]

7.
Schnitzbauer AA, Lang SA, Goessmann H, et al. Right portal vein ligation combined with in situ splitting induces rapid left lateral liver lobe hypertrophy enabling 2-staged extended right hepatic resection in small-for-size settings. Ann Surg 2012;255(3):405–14. [CrossRef]
[Pubmed]

8.
Zorzi D, Laurent A, Pawlik TM, Lauwers GY, Vauthey JN, Abdalla EK. Chemotherapy-associated hepatotoxicity and surgery for colorectal liver metastases. Br J Surg 2007;94(3):274–86. [CrossRef]
[Pubmed]

9.
Abdalla EK, Hicks ME, Vauthey JN. Portal vein embolization: Rationale, technique and future prospects. Br J Surg 2001;88(2):165–75. [CrossRef]
[Pubmed]

10.
She WH, Chok KS. Strategies to increase the resectability of hepatocellular carcinoma. World J Hepatol 2015;7(18):2147–54. [CrossRef]
[Pubmed]

11.
Shindoh J, Tzeng CWD, Vauthey JN. Portal vein embolization for hepatocellular carcinoma. Liver Cancer 2012;1(3–4):159–67. [CrossRef]
[Pubmed]

12.
May BJ, Madoff DC. Portal vein embolization: Rationale, technique, and current application. Semin Intervent Radiol 2012;29(2):81–9. [CrossRef]
[Pubmed]

13.
Loffroy R, Favelier S, Chevallier O, et al. Preoperative portal vein embolization in liver cancer: Indications, techniques and outcomes. Quant Imaging Med Surg 2015;5(5):730–9. [CrossRef]
[Pubmed]

14.
Cieslak KP, Bennink RJ, de Graaf W, et al. Measurement of liver function using hepatobiliary scintigraphy improves risk assessment in patients undergoing major liver resection. HPB (Oxford) 2016;18(9):773–80. [CrossRef]
[Pubmed]

15.
Nadalin S, Capobianco I, Li J, Girotti P, Königsrainer I, Königsrainer A. Indications and limits for associating liver partition and portal vein ligation for staged hepatectomy (ALPPS). Lessons learned from 15 cases at a single centre. Z Gastroenterol 2014;52(1):35–42. [CrossRef]
[Pubmed]

16.
Schadde E, Ardiles V, Robles-Campos R, et al. Early survival and safety of ALPPS: First report of the International ALPPS Registry. Ann Surg 2014;260(5):829–38. [CrossRef]
[Pubmed]

17.
Al Hasan I, Tun-Abraham ME, Wanis KN, et al. Optimizing associated liver partition and portal vein ligation for staged hepatectomy outcomes: Surgical experience or appropriate patient selection? Can J Surg 2017;60(6):408–15. [CrossRef]
[Pubmed]

18.
Alvarez FA, Ardiles V, Sanchez Claria R, Pekolj J, de Santibañes E. Associating liver partition and portal vein ligation for staged hepatectomy (ALPPS): Tips and tricks. J Gastrointest Surg 2013;17(4):814–21. [CrossRef]
[Pubmed]

19.
Imamura H, Shimada R, Kubota M, et al. Preoperative portal vein embolization: An audit of 84 patients. Hepatology 1999;29(4):1099–105. [CrossRef]
[Pubmed]

20.
Ko HK, Ko GY, Sung KB, Gwon DI, Yoon HK. Portal vein embolization via percutaneous transsplenic access prior to major hepatectomy for patients with insufficient future liver remnant. J Vasc Interv Radiol 2016;27(7):981–6. [CrossRef]
[Pubmed]

21.
Avritscher R, de Baere T, Murthy R, Deschamps F, Madoff DC. Percutaneous transhepatic portal vein embolization: Rationale, technique, and outcomes. Semin Intervent Radiol 2008;25(2):132–45. [CrossRef]
[Pubmed]

22.
Kinoshita H, Sakai K, Hirohashi K, Igawa S, Yamasaki O, Kubo S. Preoperative portal vein embolization for hepatocellular carcinoma. World J Surg 1986;10(5):803–8. [CrossRef]
[Pubmed]

23.
Nagino M, Nimura Y, Kamiya J, Kondo S, Kanai M. Selective percutaneous transhepatic embolization of the portal vein in preparation for extensive liver resection: The ipsilateral approach. Radiology 1996;200(2):559–63. [CrossRef]
[Pubmed]

24.
May BJ, Madoff DC. Controversies of preoperative portal vein embolization. Hepat Oncol 2016;3(2):155–66. [CrossRef]
[Pubmed]

25.
Yamakado K, Takeda K, Nishide Y, et al. Portal vein embolization with steel coils and absolute ethanol: A comparative experimental study with canine liver. Hepatology 1995;22(6):1812–8.
[Pubmed]

26.
Madoff DC, Abdalla EK, Gupta S, et al. Transhepatic ipsilateral right portal vein embolization extended to segment IV: Improving hypertrophy and resection outcomes with spherical particles and coils. J Vasc Interv Radiol 2005;16(2 Pt 1):215–25. [CrossRef]
[Pubmed]

27.
Obed A, Schnitzbauer AA, Tsui TY, et al. Living donor liver resection: A low-tech but highly efficient technique. The Regensburg experience. Langenbecks Arch Surg 2008;393(3):413–21. [CrossRef]
[Pubmed]

28.
Shoup M, Gonen M, D’Angelica M, et al. Volumetric analysis predicts hepatic dysfunction in patients undergoing major liver resection. J Gastrointest Surg 2003;7(3):325–30. [CrossRef]
[Pubmed]

29.
Ribero D, Chun YS, Vauthey JN. Standardized liver volumetry for portal vein embolization. Semin Intervent Radiol 2008;25(2):104–9. [CrossRef]
[Pubmed]

30.
Vauthey JN, Abdalla EK, Doherty DA, et al. Body surface area and body weight predict total liver volume in Western adults. Liver Transpl 2002;8(3):233–40. [CrossRef]
[Pubmed]

31.
Urata K, Kawasaki S, Matsunami H, et al. Calculation of child and adult standard liver volume for liver transplantation. Hepatology 1995;21(5):1317–21.
[Pubmed]

32.
Vauthey JN, Chaoui A, Do KA, et al. Standardized measurement of the future liver remnant prior to extended liver resection: Methodology and clinical associations. Surgery 2000;127(5):512–9. [CrossRef]
[Pubmed]

33.
Maundura M, Koea JB. Assessment and optimization of the future liver remnant. 2017. [Available at: https://www.intechopen.com/books/updates-in-liver-cancer/assessment-and-optimization-of-the-future-liver-remnant] [CrossRef]

34.
Shindoh J, Truty MJ, Aloia TA, et al. Kinetic growth rate after portal vein embolization predicts posthepatectomy outcomes: Toward zero liver-related mortality in patients with colorectal liver metastases and small future liver remnant. J Am Coll Surg 2013;216(2):201–9. [CrossRef]
[Pubmed]

35.
Uz Z, Ince C, Rassam F, Ergin B, van Lienden KP, van Gulik TM. Assessment of hepatic microvascular flow and density in patients undergoing preoperative portal vein embolization. HPB (Oxford) 2019;21(2):187–94. [CrossRef]
[Pubmed]

36.
van Lienden KP, van den Esschert JW, de Graaf W, et al. Portal vein embolization before liver resection: A systematic review. Cardiovasc Intervent Radiol 2013;36(1):25–34. [CrossRef]
[Pubmed]

37.
Rassam F, Olthof PB, van Lienden KP, et al. Functional and volumetric assessment of liver segments after portal vein embolization: Differences in hypertrophy response. Surgery 2019;165(4):686–95. [CrossRef]
[Pubmed]

38.
de Graaf W, van Lienden KP, van den Esschert JW, Bennink RJ, van Gulik TM. Increase in future remnant liver function after preoperative portal vein embolization. Br J Surg 2011;98(6):825–34. [CrossRef]
[Pubmed]

39.
Chan KS, Low JK, Shelat VG. Associated liver partition and portal vein ligation for staged hepatectomy: A review. Transl Gastroenterol Hepatol 2020;5:35. [CrossRef]
[Pubmed]

40.
Cai YL, Song PP, Tang W, Cheng NS. An updated systematic review of the evolution of ALPPS and evaluation of its advantages and disadvantages in accordance with current evidence. Medicine (Baltimore) 2016;95(24):e3941. [CrossRef]
[Pubmed]

41.
Kang D, Schadde E. Hypertrophy and liver function in ALPPS: Correlation with morbidity and mortality. Visc Med 2017;33(6):426–33. [CrossRef]
[Pubmed]

42.
Kambakamba P, Stocker D, Reiner CS, et al. Liver kinetic growth rate predicts postoperative liver failure after ALPPS. HPB (Oxford) 2016;18(10):800–5. [CrossRef]
[Pubmed]

43.
Bednarsch J, Czigany Z, Sharmeen S, et al. ALPPS versus two-stage hepatectomy for colorectal liver metastases—a comparative retrospective cohort study. World J Surg Oncol 2020;18(1):140. [CrossRef]
[Pubmed]

44.
Tanaka K, Matsuo K, Murakami T, et al. Associating liver partition and portal vein ligation for staged hepatectomy (ALPPS): Short-term outcome, functional changes in the future liver remnant, and tumour growth activity. Eur J Surg Oncol 2015;41(4):506–12. [CrossRef]
[Pubmed]

45.
Tomassini F, D’Asseler Y, Linecker M, et al. Hepatobiliary scintigraphy and kinetic growth rate predict liver failure after ALPPS: A multi-institutional study. HPB (Oxford) 2020;22(10):1420–8. [CrossRef]
[Pubmed]

46.
Serenari M, Zanello M, Schadde E, et al. Importance of primary indication and liver function between stages: Results of a multicenter Italian audit of ALPPS 2012–2014. HPB (Oxford) 2016;18(5):419–27. [CrossRef]
[Pubmed]

47.
de Graaf W, van Lienden KP, Dinant S, et al. Assessment of future remnant liver function using hepatobiliary scintigraphy in patients undergoing major liver resection. J Gastrointest Surg 2010;14(2):369–78. [CrossRef]
[Pubmed]

48.
Olthof PB, Tomassini F, Huespe PE, et al. Hepatobiliary scintigraphy to evaluate liver function in associating liver partition and portal vein ligation for staged hepatectomy: Liver volume overestimates liver function. Surgery 2017;162(4):775–83. [CrossRef]
[Pubmed]

49.
Chan A, Zhang WY, Chok K, et al. ALPPS versus portal vein embolization for hepatitis-related hepatocellular carcinoma: A changing paradigm in modulation of future liver remnant before major hepatectomy. Ann Surg 2019. [CrossRef]
[Pubmed]

50.
Croome KP, Hernandez-Alejandro R, Parker M, Heimbach J, Rosen C, Nagorney DM. Is the liver kinetic growth rate in ALPPS unprecedented when compared with PVE and living donor liver transplant? A multicentre analysis. HPB (Oxford) 2015;17(6):477–84. [CrossRef]
[Pubmed]

51.
Cieslak KP, Huisman F, Bais T, et al. Future remnant liver function as predictive factor for the hypertrophy response after portal vein embolization. Surgery 2017;162(1):37–47. [CrossRef]
[Pubmed]

52.
Eshmuminov D, Raptis DA, Linecker M, Wirsching A, Lesurtel M, Clavien PA. Meta-analysis of associating liver partition with portal vein ligation and portal vein occlusion for two-stage hepatectomy. Br J Surg 2016;103(13):1768–82. [CrossRef]
[Pubmed]

53.
Liu Y, Yang Y, Gu S, Tang K. A systematic review and meta-analysis of associating liver partition and portal vein ligation for staged hepatectomy (ALPPS) versus traditional staged hepatectomy. Medicine (Baltimore) 2019;98(15):e15229. [CrossRef]
[Pubmed]

54.
Matsuo K, Murakami T, Kawaguchi D, et al. Histologic features after surgery associating liver partition and portal vein ligation for staged hepatectomy versus those after hepatectomy with portal vein embolization. Surgery 2016;159(5):1289–98. [CrossRef]
[Pubmed]

55.
Sparrelid E, Jonas E, Tzortzakakis A, et al. Dynamic evaluation of liver volume and function in associating liver partition and portal vein ligation for staged hepatectomy. J Gastrointest Surg 2017;21(6):967–74. [CrossRef]
[Pubmed]

56.
Farges O, Belghiti J, Kianmanesh R, et al. Portal vein embolization before right hepatectomy: Prospective clinical trial. Ann Surg 2003;237(2):208–17. [CrossRef]
[Pubmed]

57.
Huiskens J, Olthof PB, van der Stok EP, et al. Does portal vein embolization prior to liver resection influence the oncological outcomes – A propensity score matched comparison. Eur J Surg Oncol 2018;44(1):108–14. [CrossRef]
[Pubmed]

58.
Wicherts DA, de Haas RJ, Andreani P, et al. Impact of portal vein embolization on long-term survival of patients with primarily unresectable colorectal liver metastases. Br J Surg 2010;97(2):240–50. [CrossRef]
[Pubmed]

59.
Ironside N, Bell R, Bartlett A, McCall J, Powell J, Pandanaboyana S. Systematic review of perioperative and survival outcomes of liver resections with and without preoperative portal vein embolization for colorectal metastases. HPB (Oxford) 2017;19(7):559–66. [CrossRef]
[Pubmed]

60.
Dindo D, Demartines N, Clavien PA. Classification of surgical complications: A new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 2004;240(2):205–13. [CrossRef]
[Pubmed]

61.
Hernandez-Alejandro R, Bertens KA, Pineda-Solis K, Croome KP. Can we improve the morbidity and mortality associated with the associating liver partition with portal vein ligation for staged hepatectomy (ALPPS) procedure in the management of colorectal liver metastases? Surgery 2015;157(2):194–201. [CrossRef]
[Pubmed]

62.
Oldhafer KJ, Donati M, Jenner RM, Stang A, Stavrou GA. ALPPS for patients with colorectal liver metastases: Effective liver hypertrophy, but early tumour recurrence. World J Surg 2014;38(6):1504–9. [CrossRef]
[Pubmed]

63.
Li J, Girotti P, Königsrainer I, Ladurner R, Königsrainer A, Nadalin S. ALPPS in right trisectionectomy: A safe procedure to avoid postoperative liver failure? J Gastrointest Surg 2013;17(5):956–61. [CrossRef]
[Pubmed]

64.
Ratti F, Cipriani F, Gagliano A, Catena M, Paganelli M, Aldrighetti L. Defining indications to ALPPS procedure: Technical aspects and open issues. Updates Surg 2014;66(1):41–9. [CrossRef]
[Pubmed]

65.
Schadde E, Raptis DA, Schnitzbauer AA, et al. Prediction of mortality after ALPPS Stage-1: An analysis of 320 patients from the International ALPPS registry. Ann Surg 2015;262(5):780–6. [CrossRef]
[Pubmed]

66.
Dokmak S, Belghiti J. Which limits to the “ALPPS” approach? Ann Surg 2012;256(3):e6. [CrossRef]
[Pubmed]

67.
López-López V, Robles-Campos R, Brusadin R, et al. Tourniquet-ALPPS is a promising treatment for very large hepatocellular carcinoma and intrahepatic cholangiocarcinoma. Oncotarget 2018;9(46):28267–80. [CrossRef]
[Pubmed]

68.
Pekolj J, Alvarez FA, Biagiola D, Villegas L, Ardiles V, de Santibañes E. Totally laparoscopic mini- ALPPS using a novel approach of laparoscopicassisted transmesenteric portal vein embolization. J Laparoendosc Adv Surg Tech A 2018;28(10):1229– 33. [CrossRef]
[Pubmed]

69.
Surjan RC, Makdissi FF, Basseres T, et al. First totally laparoscopic ALPPS procedure with selective hepatic artery clamping: Case report of a new technique. Medicine (Baltimore) 2016;95(29):e4236. [CrossRef]
[Pubmed]

70.
Petrowsky H, Györi G, de Oliveira M, Lesurtel M, Clavien PA. Is partial-ALPPS safer than ALPPS? A single-center experience. Ann Surg 2015;261(4):e90– 2. [CrossRef]
[Pubmed]

71.
Shindoh J, Vauthey JN, Zimmitti G, et al. Analysis of the efficacy of portal vein embolization for patients with extensive liver malignancy and very low future liver remnant volume, including a comparison with the associating liver partition with portal vein ligation for staged hepatectomy approach. J Am Coll Surg 2013;217(1):126–34. [CrossRef]
[Pubmed]

72.
Björnsson B, Sparrelid E, Røsok B, et al. Associating liver partition and portal vein ligation for staged hepatectomy in patients with colorectal liver metastases–Intermediate oncological results. Eur J Surg Oncol 2016;42(4):531–7. [CrossRef]
[Pubmed]

73.
Simoneau E, Hassanain M, Shaheen M, et al. Portal vein embolization and its effect on tumour progression for colorectal cancer liver metastases. Br J Surg 2015;102(10):1240–9. [CrossRef]
[Pubmed]

74.
Hoekstra LT, van Lienden KP, Doets A, Busch ORC, Gouma DJ, van Gulik TM. Tumour progression after preoperative portal vein embolization. Ann Surg 2012;256(5):812–8. [CrossRef]
[Pubmed]

75.
Giglio MC, Giakoustidis A, Draz A, et al. Oncological outcomes of major liver resection following portal vein embolization: A systematic review and meta-analysis. Ann Surg Oncol 2016;23(11):3709–17. [CrossRef]
[Pubmed]

76.
Fischer C, Melstrom LG, Arnaoutakis D, et al. Chemotherapy after portal vein embolization to protect against tumour growth during liver hypertrophy before hepatectomy. JAMA Surg 2013;148(12):1103–8. [CrossRef]
[Pubmed]

77.
Moris D, Ronnekleiv-Kelly S, Kostakis ID, et al. Operative results and oncologic outcomes of associating liver partition and portal vein ligation for staged hepatectomy (ALPPS) versus two-stage hepatectomy (TSH) in patients with unresectable colorectal liver metastases: A systematic review and meta-analysis. World J Surg 2018;42(3):806–15. [CrossRef]
[Pubmed]

78.
Ratti F, Schadde E, Masetti M, et al. Strategies to increase the resectability of patients with colorectal liver metastases: A multi-center case-match analysis of ALPPS and conventional two-stage hepatectomy. Ann Surg Oncol 2015;22(6):1933–42. [CrossRef]
[Pubmed]

79.
Adam R, Imai K, Benitez CC, et al. Outcome after associating liver partition and portal vein ligation for staged hepatectomy and conventional two-stage hepatectomy for colorectal liver metastases. Br J Surg 2016;103(11):1521–9. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Jeremy Kay Hock Lee - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Kenneth Jun Chuen Low - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Bryant Po Yuen Lim - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Lim Ai Ying Hannah - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guarantor of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2021 Jeremy Kay Hock Lee et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.